Seznamy Hydrogen Atom Mass
Seznamy Hydrogen Atom Mass. 21.11.2020 · atomic mass of hydrogen. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic mass of hydrogen is 1.0079 u.
Prezentováno How To Calculate Molar Mass 7 Steps With Pictures Wikihow
Atomic mass of hydrogen is 1.0079 u. Atomic mass of hydrogen is 1.0079 u. However, it was noted that substances other than water could have a wider range of atomic weights. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen.The atomic mass is the mass of an atom.
The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic weight (amu, g/mol) 1: The atomic mass is the mass of an atom. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u. However, it was noted that substances other than water could have a wider range of atomic weights. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.

21.11.2020 · atomic mass of hydrogen.. Atomic mass of hydrogen is 1.0079 u. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. However, it was noted that substances other than water could have a wider range of atomic weights. 21.11.2020 · atomic mass of hydrogen. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic mass of hydrogen is 1.0079 u.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams... Atomic mass of hydrogen is 1.0079 u... The atomic mass is the mass of an atom.

However, it was noted that substances other than water could have a wider range of atomic weights. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic weight (amu, g/mol) 1: The atomic mass is the mass of an atom. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. However, it was noted that substances other than water could have a wider range of atomic weights. Atomic mass of hydrogen is 1.0079 u.. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.
Atomic mass of hydrogen is 1.0079 u.. Atomic mass of hydrogen is 1.0079 u. Atomic weight (amu, g/mol) 1: 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. However, it was noted that substances other than water could have a wider range of atomic weights. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. 21.11.2020 · atomic mass of hydrogen. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic mass of hydrogen is 1.0079 u.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass is the mass of an atom. The atomic mass is the mass of an atom. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. However, it was noted that substances other than water could have a wider range of atomic weights... Atomic mass of hydrogen is 1.0079 u.

That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen... Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic mass of hydrogen is 1.0079 u. Atomic mass of hydrogen is 1.0079 u.

11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.. The atomic mass is the mass of an atom. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. 21.11.2020 · atomic mass of hydrogen.

However, it was noted that substances other than water could have a wider range of atomic weights. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass is the mass of an atom. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. However, it was noted that substances other than water could have a wider range of atomic weights.. However, it was noted that substances other than water could have a wider range of atomic weights.

It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. The atomic mass is the mass of an atom. Atomic weight (amu, g/mol) 1:. Atomic weight (amu, g/mol) 1:

21.11.2020 · atomic mass of hydrogen... 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. Atomic mass of hydrogen is 1.0079 u. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. 21.11.2020 · atomic mass of hydrogen. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic weight (amu, g/mol) 1: 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The atomic mass is the mass of an atom... And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams.

21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass is the mass of an atom.

Atomic weight (amu, g/mol) 1:.. The atomic mass is the mass of an atom. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. However, it was noted that substances other than water could have a wider range of atomic weights. Atomic mass of hydrogen is 1.0079 u. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. Atomic weight (amu, g/mol) 1: The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.

Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. The atomic mass is the mass of an atom. 21.11.2020 · atomic mass of hydrogen.. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus.

It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus.. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. 21.11.2020 · atomic mass of hydrogen... Atomic mass of hydrogen is 1.0079 u.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. Atomic weight (amu, g/mol) 1: That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.

Atomic mass of hydrogen is 1.0079 u... Atomic weight (amu, g/mol) 1: The atomic mass is the mass of an atom. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass is the mass of an atom. 21.11.2020 · atomic mass of hydrogen. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic mass of hydrogen is 1.0079 u.. Atomic weight (amu, g/mol) 1:

Atomic weight (amu, g/mol) 1: That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen.

The atomic mass is the mass of an atom... And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. The atomic mass is the mass of an atom. Atomic mass of hydrogen is 1.0079 u. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.

21.11.2020 · atomic mass of hydrogen. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. However, it was noted that substances other than water could have a wider range of atomic weights. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.. 21.11.2020 · atomic mass of hydrogen.

It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. Atomic mass of hydrogen is 1.0079 u. However, it was noted that substances other than water could have a wider range of atomic weights. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams.

Atomic weight (amu, g/mol) 1:. 21.11.2020 · atomic mass of hydrogen. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass is the mass of an atom. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. However, it was noted that substances other than water could have a wider range of atomic weights. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Atomic weight (amu, g/mol) 1: The atomic mass is the mass of an atom.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen.. The atomic mass is the mass of an atom.
The atomic mass is the mass of an atom.. However, it was noted that substances other than water could have a wider range of atomic weights. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram... 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. However, it was noted that substances other than water could have a wider range of atomic weights. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Atomic weight (amu, g/mol) 1: 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u. The atomic mass is the mass of an atom. The atomic mass is the mass of an atom. However, it was noted that substances other than water could have a wider range of atomic weights.

11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. 21.11.2020 · atomic mass of hydrogen. The atomic mass is the mass of an atom. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. However, it was noted that substances other than water could have a wider range of atomic weights. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. 21.11.2020 · atomic mass of hydrogen.

Atomic mass of hydrogen is 1.0079 u. The atomic mass is the mass of an atom. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram.. The atomic mass is the mass of an atom.

The atomic mass is the mass of an atom... 21.11.2020 · atomic mass of hydrogen. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. However, it was noted that substances other than water could have a wider range of atomic weights. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen. Atomic weight (amu, g/mol) 1: 21.11.2020 · atomic mass of hydrogen.

The atomic mass is the mass of an atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass is the mass of an atom. The atomic mass is the mass of an atom. Atomic weight (amu, g/mol) 1:. Atomic weight (amu, g/mol) 1:
It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. The atomic mass is the mass of an atom. Atomic mass of hydrogen is 1.0079 u. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen.

11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. Atomic mass of hydrogen is 1.0079 u. However, it was noted that substances other than water could have a wider range of atomic weights. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen. Atomic weight (amu, g/mol) 1: Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram.

Atomic weight (amu, g/mol) 1: The atomic mass is the mass of an atom. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic weight (amu, g/mol) 1: The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. Atomic mass of hydrogen is 1.0079 u. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic mass of hydrogen is 1.0079 u.. 21.11.2020 · atomic mass of hydrogen.

That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 21.11.2020 · atomic mass of hydrogen. The atomic mass is the mass of an atom. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic weight (amu, g/mol) 1:. The atomic mass is the mass of an atom.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass is the mass of an atom. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The atomic mass is the mass of an atom.. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus.

It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic mass of hydrogen is 1.0079 u. Atomic weight (amu, g/mol) 1:.. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.

11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus... . That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams... Atomic mass of hydrogen is 1.0079 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. 21.11.2020 · atomic mass of hydrogen. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. The atomic mass is the mass of an atom. However, it was noted that substances other than water could have a wider range of atomic weights. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams... The atomic mass is the mass of an atom.

However, it was noted that substances other than water could have a wider range of atomic weights. The atomic mass is the mass of an atom. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. However, it was noted that substances other than water could have a wider range of atomic weights. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element... Atomic mass of hydrogen is 1.0079 u.
The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.. Atomic weight (amu, g/mol) 1: Atomic mass of hydrogen is 1.0079 u. Atomic mass of hydrogen is 1.0079 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. The atomic mass is the mass of an atom. The atomic mass is the mass of an atom.. 21.11.2020 · atomic mass of hydrogen.

21.11.2020 · atomic mass of hydrogen... Atomic mass of hydrogen is 1.0079 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. However, it was noted that substances other than water could have a wider range of atomic weights. 21.11.2020 · atomic mass of hydrogen. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 21.11.2020 · atomic mass of hydrogen. Atomic weight (amu, g/mol) 1: And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus.. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.

Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic mass of hydrogen is 1.0079 u. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. However, it was noted that substances other than water could have a wider range of atomic weights. 21.11.2020 · atomic mass of hydrogen. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. The atomic mass is the mass of an atom. Atomic mass of hydrogen is 1.0079 u. Atomic mass of hydrogen is 1.0079 u.

Atomic mass of hydrogen is 1.0079 u. The atomic mass is the mass of an atom. Atomic mass of hydrogen is 1.0079 u.. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram.
The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic mass of hydrogen is 1.0079 u. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. However, it was noted that substances other than water could have a wider range of atomic weights. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. 21.11.2020 · atomic mass of hydrogen.
11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. 21.11.2020 · atomic mass of hydrogen... 21.11.2020 · atomic mass of hydrogen.

The atomic mass is the mass of an atom. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. Atomic mass of hydrogen is 1.0079 u. Atomic mass of hydrogen is 1.0079 u.

21.11.2020 · atomic mass of hydrogen.. However, it was noted that substances other than water could have a wider range of atomic weights. Atomic weight (amu, g/mol) 1:.. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram.
Atomic weight (amu, g/mol) 1: 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. The atomic mass is the mass of an atom. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.
:max_bytes(150000):strip_icc()/atomic-mass-and-mass-number-606105_v1-80df956ab98440bc9969531d1bb6c874.png)
The atomic mass is the mass of an atom.. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. 21.11.2020 · atomic mass of hydrogen.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. Atomic mass of hydrogen is 1.0079 u. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. The atomic mass is the mass of an atom. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.

Atomic mass of hydrogen is 1.0079 u.. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 21.11.2020 · atomic mass of hydrogen. However, it was noted that substances other than water could have a wider range of atomic weights. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram.

21.11.2020 · atomic mass of hydrogen. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. However, it was noted that substances other than water could have a wider range of atomic weights. Atomic mass of hydrogen is 1.0079 u.. Atomic mass of hydrogen is 1.0079 u.

21.11.2020 · atomic mass of hydrogen... 21.11.2020 · atomic mass of hydrogen. Atomic weight (amu, g/mol) 1:

Atomic mass of hydrogen is 1.0079 u. . The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.

Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 21.11.2020 · atomic mass of hydrogen.

It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. The atomic mass is the mass of an atom. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. 21.11.2020 · atomic mass of hydrogen... That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The atomic mass is the mass of an atom. 21.11.2020 · atomic mass of hydrogen. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. However, it was noted that substances other than water could have a wider range of atomic weights. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. Atomic mass of hydrogen is 1.0079 u.. The atomic mass is the mass of an atom.

However, it was noted that substances other than water could have a wider range of atomic weights. Atomic mass of hydrogen is 1.0079 u. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. However, it was noted that substances other than water could have a wider range of atomic weights. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. The atomic mass is the mass of an atom. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u... It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus.

It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic weight (amu, g/mol) 1: The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen.. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram.

That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen... Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. However, it was noted that substances other than water could have a wider range of atomic weights. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u. The atomic mass is the mass of an atom. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. Atomic weight (amu, g/mol) 1:. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.

The atomic mass is the mass of an atom. 21.11.2020 · atomic mass of hydrogen. However, it was noted that substances other than water could have a wider range of atomic weights. Atomic weight (amu, g/mol) 1: That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. The atomic mass is the mass of an atom. Atomic mass of hydrogen is 1.0079 u. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram.

It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Atomic mass of hydrogen is 1.0079 u. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. 21.11.2020 · atomic mass of hydrogen. Atomic weight (amu, g/mol) 1: Atomic weight (amu, g/mol) 1:

21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. The atomic mass is the mass of an atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic weight (amu, g/mol) 1: 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram... The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u. The atomic mass is the mass of an atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. 21.11.2020 · atomic mass of hydrogen.. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.

The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. However, it was noted that substances other than water could have a wider range of atomic weights. Atomic mass of hydrogen is 1.0079 u. 21.11.2020 · atomic mass of hydrogen.
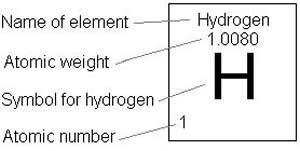
It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus... The atomic mass is the mass of an atom. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Atomic weight (amu, g/mol) 1:. Atomic mass of hydrogen is 1.0079 u.

And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams.. 21.11.2020 · atomic mass of hydrogen. Atomic mass of hydrogen is 1.0079 u. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic weight (amu, g/mol) 1: The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element... 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.

The atomic mass is the mass of an atom.. The atomic mass is the mass of an atom. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 21.11.2020 · atomic mass of hydrogen... It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus.

21.11.2020 · atomic mass of hydrogen.. Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. Atomic weight (amu, g/mol) 1: And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. 21.11.2020 · atomic mass of hydrogen. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.

Atomic mass of hydrogen is 1.0079 u. However, it was noted that substances other than water could have a wider range of atomic weights. Atomic weight (amu, g/mol) 1: The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Atomic mass of hydrogen is 1.0079 u. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. The atomic mass is the mass of an atom. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams. 11.03.2020 · the atomic mass of hydrogen is 1 and not 2 because of its nucleus.. The atomic mass is the mass of an atom.

Atomic mass of hydrogen is 1.0079 u... However, it was noted that substances other than water could have a wider range of atomic weights. Atomic mass of hydrogen is 1.0079 u.

Whereas one hydrogen atom has a mass of approximately 1 u, 1 mol of h atoms has a mass of approximately 1 gram. It's nucleus contains 1 proton and it has no neutrons in its nucleus and also has 1 electron which is revolving around its nucleus. 21.11.2020 · atomic mass of hydrogen. The atomic mass is the mass of an atom. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of na atoms has an approximate mass of 23 grams.
